IntraCellular Diagnostics, Inc®
|
|
|
IntraCellular Diagnostics, Inc.®
Analytical Scanning
Electron Microscopy and Nano Analysis
Office based 60 seconds, non-invasive, specimen collection
Direct Testing For Vital Mineral Electrolytes In The Cell:
Order a Specimen Collection Kit
Order a
Specimen 60
Second Specimen
Published
Research &
|
The Power Mineral Magnesium
Occurrence in Nature
The chlorophyll molecule
The pigments in leaves and blood are chemically related. Even when at first sight it seems unbelievable: i.e. the pigment in leaves and red blood corpuscles actually have something in common. Both, chlorophyll and hemoglobin are so-called porphyrin rings. The only difference is that the central atom
in hemoglobin is iron, whereas in chlorophyll it is magnesium. The technical information in this web site is not intended to recommend treatment or make specific diagnosis based on such data. It is intended for professional informational purposes, (from current medical literature), to assist practitioners in choosing appropriate protocols and modalities. Decisions on patient care should be based on all laboratory tests, health histories and clinical evaluations.
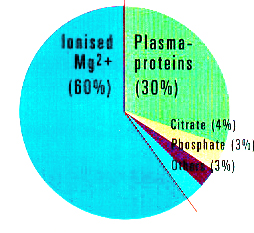
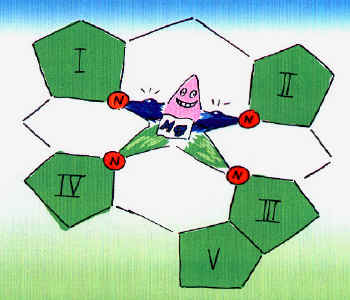
Magnesium Balance Mg2+reserves in the body Bones about 60%, Cells about 40% Interstitial compartment and Serum About 1% Fig. 1: Mg2+Supply. Intake with food / Absorption quotient 30 - 70%. Fig. 2. Renal elimination: 100 mg. per day. Fig. 3. Mg2+Reserves in the body:
3.
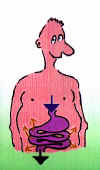
Magnesium circulation within the body... Begins with the intake of food - we cannot produce the valuable mineral alone. After ingestion with food, the major part of the mineral has initially a good way to travel: to the small intestine. Only then is it absorbed:
Important! Interaction with iron when supplements are used: the absorption of magnesium interferes with that of iron! In replacement therapy with both minerals, they should be taken at a 2 to 3 hour interval from one another.
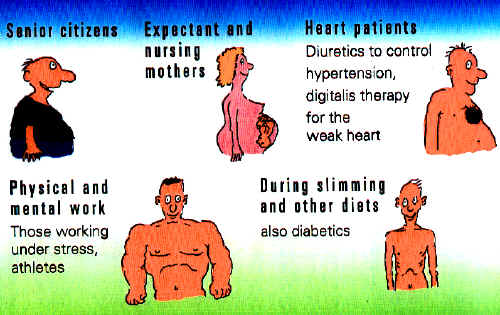
What does the body do with this valuable substance? 60% of magnesium is stored in bone. Bone forms our most important stocks of magnesium and the body can call on two thirds of these stores (as 45% of the total reserves) if need be. 40% migrates into soft tissue, principally muscles and organs. Magnesium is the second most frequently occurring intracellular cation and is found mainly in the cells. Extracellular, i.e. in the interstitial fluid between the cells, and in blood serum, only 1% of magnesium is to be found. The major part is ionized and therefore pharmacologically active and a smaller portion is bound to other substances, to citrate, for example. The nominal value for magnesium reserves within the body is approximately 24 to 28 grams. Magnesium is excreted via the intestines and the kidneys: Only approximately 5% of the amount filtered by the kidneys is ultimately excreted. The major part returns to the blood vessels via the ascending limb of Henle's loop.
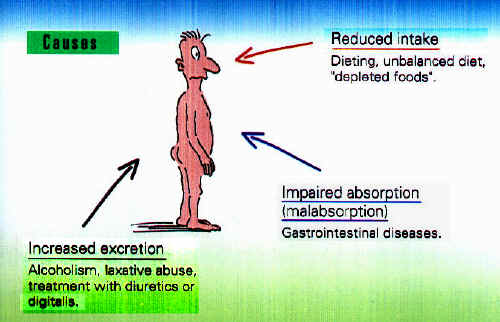
Important! Side-effect of loop diuretics: these exert their action precisely at this site and in so doing, block re-absorption of magnesium. Long-term diuretic treatment therefore frequently causes magnesium deficiency!
Reported Magnesium Requirements
During lactation also, magnesium replacement
therapy is recommended. In this case, the requirements increase to 375 mg per day. In
foods, the magnesium content is varied in its order of magnitude from one food to another.
Therefore, believe it or not, 100 g of sunflower weeds for example contain 420 mg of
magnesium. A breakfast of sunflower seed bread is therefore just what the doctor ordered!
Magnesium Content
In foods, the magnesium content is varied in its order of magnitude from one food to another. Therefore, believe it or not, 100g of sunflower seeds for example contain 350 to 420 mg of magnesium. A breakfast of sunflower seed bread is therefore just what the doctor ordered! Muesli enthusiasts settle for wheatgerm (308 mg per 100g), soup fans for white beans (130 mg per 100 g). Also, with a good deal of imagination, a glass of beer can be justified by the need to supply magnesium. A half litre contains at any rate 45 mg of magnesium. None the less, there are symptoms of
magnesium deficiency. These result, among other things, from acid rain!
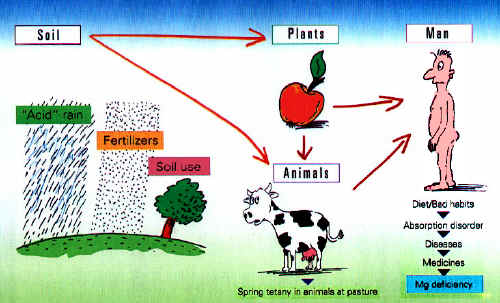
The Origins of Magnesium Deficiency From acid rain to magnesium deficiency Acid rain not only damages forests, but also the soil. Fertilizers become necessary - yet fertilizer often contains too little magnesium. Soils poor in magnesium are the result. Animals suffer: the tetany occurring early in the
year in grazing animals is not infrequently fatal without magnesium
injections. Humans also suffer depletion as a result because soil products
or foods contain too little magnesium.
Magnesium Deficiency Magnesium deficiency in humans During all the phases of magnesium circulation within the body, there are factors which may lead of magnesium deficiency:
Increased Magnesium Requirements According to age and living conditions, increased magnesium requirements may occur in humans. We have already mentioned the greater need during lactation and fasting diets. Magnesium replacement therapy may also, however, become necessary in individuals placed under professional stress or practicing active sport. In addition, magnesium is also employed in cardiac patients, in order to compensate for the increased magnesium excretion under diuretics or digitalis treatment. The technical information in this web site
is not intended to recommend treatment or make specific diagnosis based on such data. It
is intended for professional informational purposes, (from current medical literature), to
assist practitioners in choosing appropriate protocols and modalities. Decisions on
patient care should be based on all laboratory tests, health histories and clinical
evaluations.
The Complex Symptoms of Mineral Deficiency and Imbalance
IntraCellular Diagnostics, Inc. 945 Town Centre DR, Suite A Medford, OR 97504 Tel. 541-245-3212 Email us:
©Copyright IntraCellular Diagnostics, Inc® 2014. All rights reserved. |



 2.
2.